▼▼▼▼▼▼▼▼▼▼▼▼
|
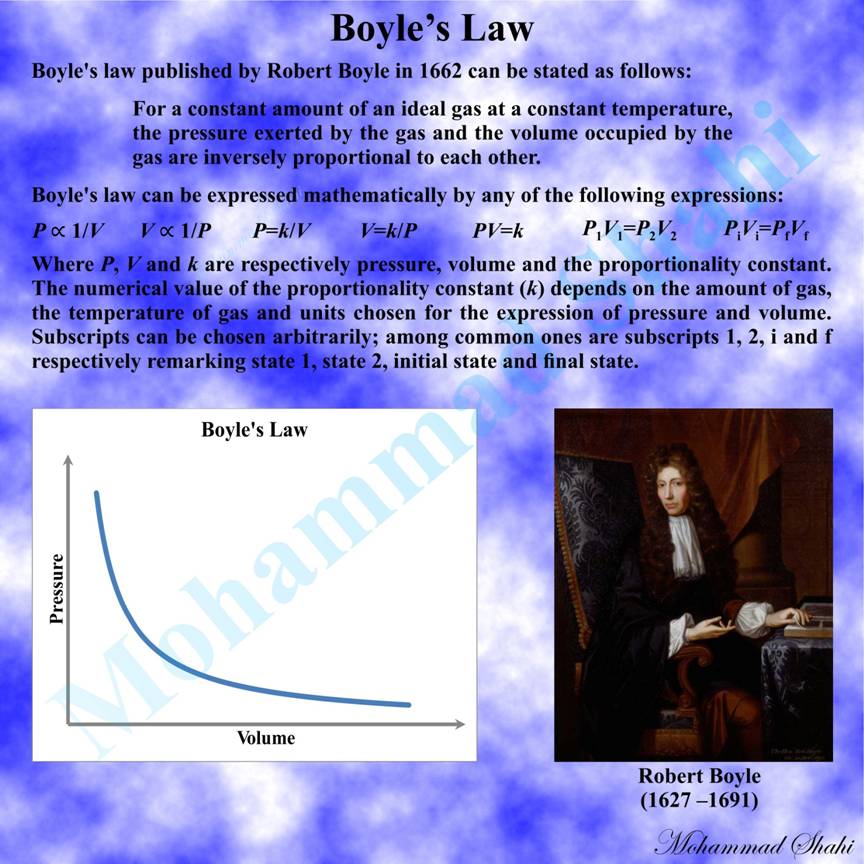
Boyle’s Law
Boyle’s law addresses the inversely proportional relationship between the pressure and volume of a gas under controlled conditions. It can be stated as follows: For a constant amount of an ideal gas at a constant temperature, the pressure exerted by the gas and the volume occupied by the gas are inversely proportional to each other.
This law was published by Robert Boyle in 1662. Later, in 1679, independently the French physicist Edme Mariotte discovered this law and also found that air volume changes with temperature. As a result, this law is sometimes found as Mariotte's law or the Boyle–Mariotte law in the literature. It may be interesting to know that Boyle’s law was the first physical law that was expressed in the form of an equation to describe the dependence of two variable quantities.
|


