▼▼▼▼▼▼▼▼▼▼▼▼
|
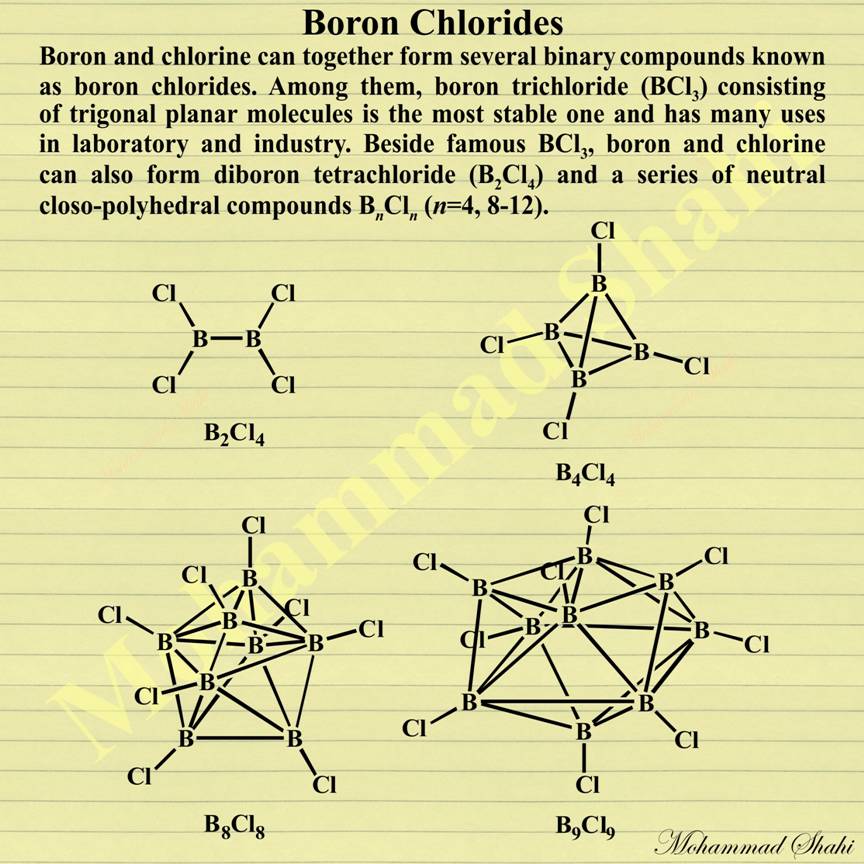
Boron Chlorides
Boron chlorides are binary compounds of boron and chlorine. The most stable one is boron trichloride (BCl3) which has trigonal planar molecules. Boron trichloride has many applications in laboratory and industry. In the laboratory, it is produced by reaction of boron trifluoride (BF3) with aluminium trichloride (AlCl3). Industrial production of boron trichloride uses direct chlorination of boron oxide and carbon at 500 °C: B2O3 + 3 C + 3 Cl2 → 2 BCl3 + 3 CO Among famous industrial applications of boron trichloride are its use in the production of elemental boron and in refining of aluminium, magnesium, zinc, and copper alloys to remove nitrides, carbides, and oxides from molten metal.
Heating boron trichloride with copper metal can produce another of boron chlorides, diboron tetrachloride (B2Cl4), via the following reaction: 2 BCl3 + 2 Cu → B2Cl4 + 2 CuCl Colourless diboron tetrachloride has planar molecules in the solid state but their structure changes to a staggered one in the gas state. It decomposes at room temperatures to give a series of neutral closo-polyhedral compounds BnCln (n=4, 8-12).
|


