▼▼▼▼▼▼▼▼▼▼▼▼
|
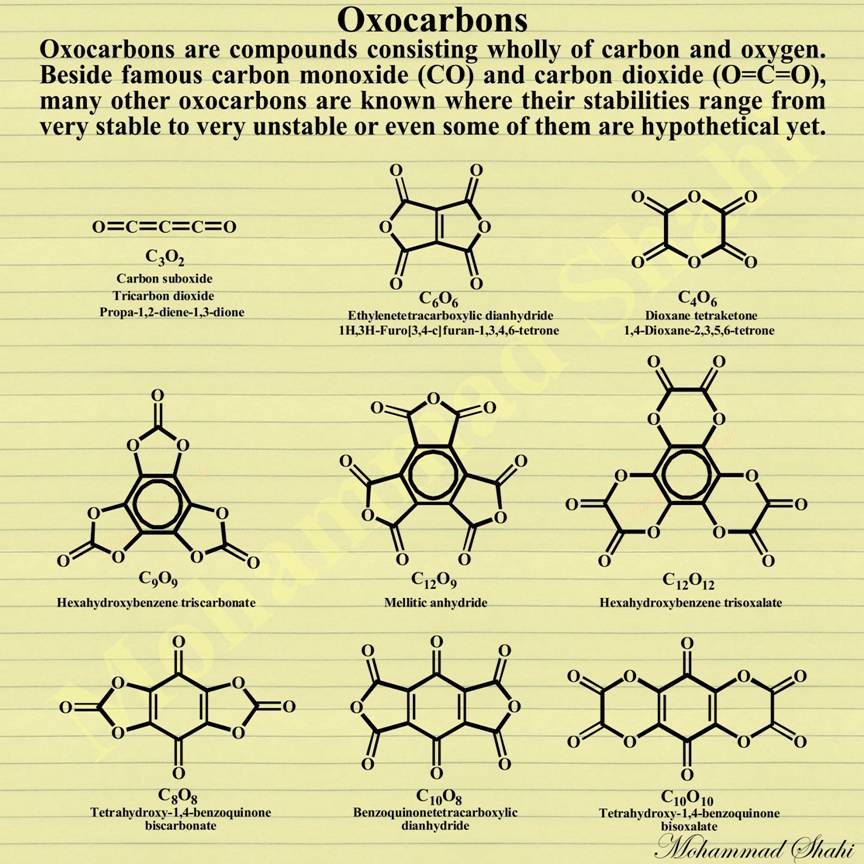
Oxocarbons
Oxocarbons are compounds consisting wholly of carbon and oxygen. We probably have already seen some of notable ones like carbon monoxide (CO), carbon dioxide (CO2), carbon suboxide (C3O2) and the mellitic anhydride (C12O9), but there exist many other ones although most of them are unstable or metastable. For example, dicarbon monoxide radical (:C=C=O), carbon trioxide (CO3), carbon tetroxide (CO4), carbon pentoxide (CO5), carbon hexoxide (CO6) and 1,2-dioxetanedione (C2O4) are among reactive carbon oxides found only in extreme situations. There also exist hypothetical ones like oxalic anhydride (C2O3 or O=(C2O)=O) and ethylene dione (C2O2 or O=C=C=O) that have been studied theoretically but haven’t been observed practically. Also among interesting ones, we have linear or cyclic polymers of carbon monoxide (-CO-)n (polyketones) and linear or cyclic polymers of carbon dioxide (-CO2-)n, such as the dimer 1,3-dioxetanedione (C2O4) and the trimer 1,3,5-trioxanetrione (C3O6) where they are also among either hypothetical ones or unstable synthesized ones except for cases like carbon monoxide or carbon dioxide themselves. Many new stable or metastable oxides have been synthesized since the 1960s, such as: C10O8 (Hammond, 1963), C6O6 (Sauer and others, 1967), C12O12 and C10O10 (Verter and others, 1968), C8O8 and C9O9 (Nallaiah, 1984), C24O6 (Rubin and others, 1990), C4O6 (Strazzolini and others, 1998).
|


